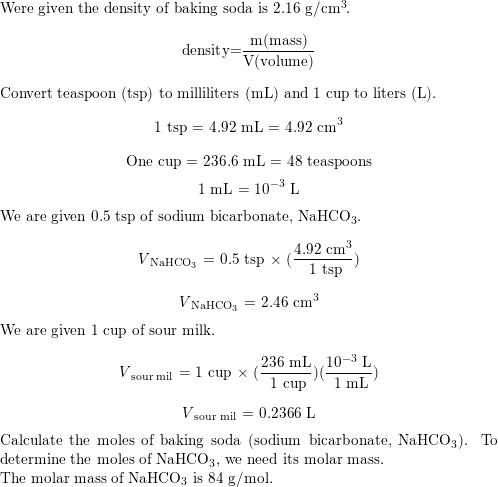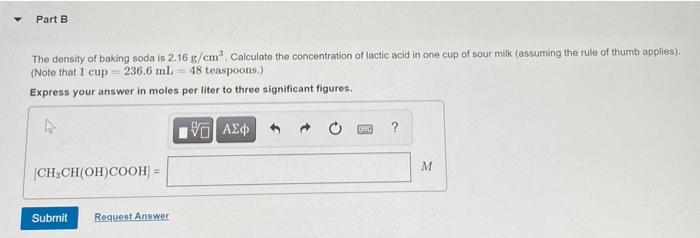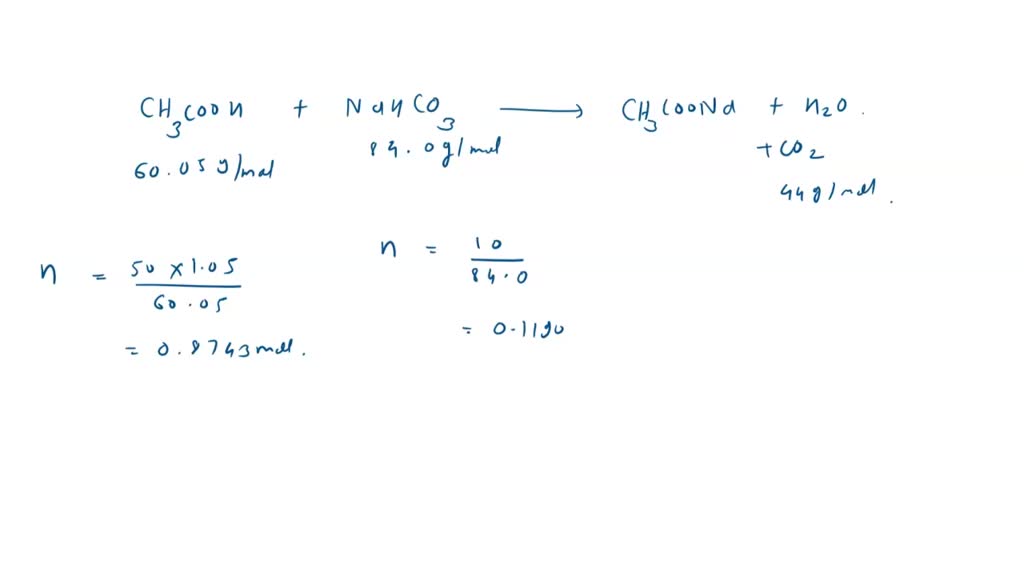
SOLVED: 10g of baking soda (Sodium Bicarbonate) is added to 50mL of vinegar (Density = 1.05g/mL). The resulting solution weighs 60.74g. How many liters of CO2 gas are released if it has

SOLVED: How many moles of baking soda are contributed by the 1/3 tsp. of baking soda in this recipe? Assume that the density of baking soda is 1 g/ml and the molecular

Chem Teaching ➿ || Chemistry on Instagram: "Sodium bicarbonate, commonly known as baking soda or bicarbonate of soda, is a chemical compound with the formula NaHCO₃. It is a salt composed of

SOLVED: Determine the mass of baking soda (84 g/mol) needed to completely react with 355 mL of vinegar. Assume vinegar is 5% (m/m) acetic acid (60 g/mol) and the density of vinegar

Preparation and properties of sodium bicarbonate‐incorporated LDPE films with deodorizing function for kimchi packaging - Jeong - 2017 - Packaging Technology and Science - Wiley Online Library

SOLVED: 6.00 cm³ of baking soda, 150 g of iron, weighs 13.2 g. The density of iron is 3.00 g/cm³. The density of white pine is 0.40 g/cm³. 43.2 g of table

Water density science experiment || Water density changes when other substances dissolve in it - YouTube